Suche:
- # Artistry
- # Biology
- # Chemistry
- # Ecological
- # Economy
- # English
- # Foreign Language
- # Geography
- # German
- # Health
- # History
- # Informatik
- # Latin
- # Mathematics
- # Media Education
- # Music
- # Physics
- # Politics / Civics
- # Preschool
- # Primary School
- # Religion
- # Society
- # Sports
- # Technology
- # Training of Teachers
- # Vocational Education
Feuer und Flamme
Zehn interaktive H5P-Aufgaben, die den Ablauf einer Schulstunde nachvollziehen, erklären in Videos Verbrennungsreaktionen und sichern das Wissen mit Aufgaben ab.
Learn moreSaure und alkalische Lösungen im Alltag
Zehn interaktive H5P-Aufgaben, die den Ablauf einer Schulstunde nachvollziehen, erklären in Videos saure und alkalische Lösungen und sichern das Wissen mit Aufgaben ab.
Learn moreLuft und ihre Zusammensetzung
Zehn interaktive H5P-Aufgaben, die den Ablauf einer Schulstunde nachvollziehen, erklären in Videos die Zusammensetzung der Luft und sichern das Wissen mit Aufgaben ab.
Learn moreKunststoffe
Zehn interaktive H5P-Aufgaben, die den Ablauf einer Schulstunde nachvollziehen, erklären in Videos Kunststoffe und sichern das Wissen mit Aufgaben ab.
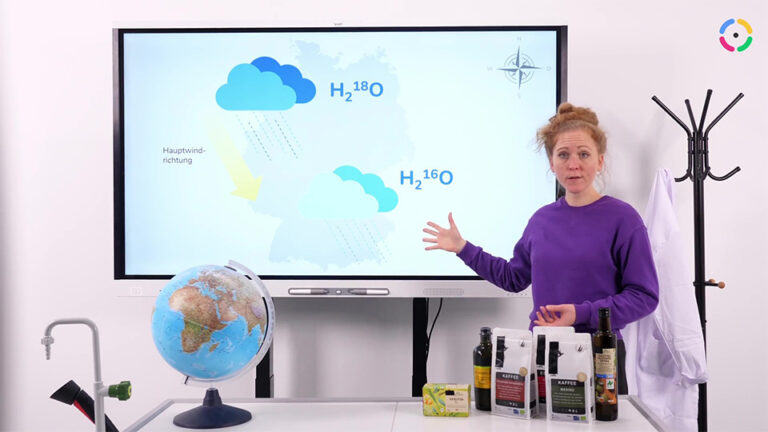
Learn moreIsotope
Zehn interaktive H5P-Aufgaben, die den Ablauf einer Schulstunde nachvollziehen, erklären in Videos Atomaufbau und Isotope und sichern das Wissen mit Aufgaben ab.
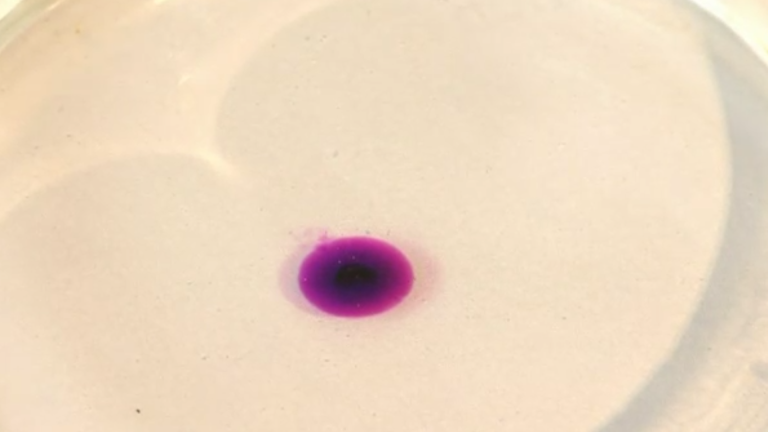
Learn moreDünnschichtchromatographie
Chromatografische Verfahren sind physikalische / chemische Trenntechniken von Stoffen.
Learn moreLerneinheit Chemie 8 – Kalk
In unserem Arbeitsheft „Lerneinheit Chemie 8 – Kalk“ finden Sie 10 interaktive und didaktisch aufbereitete Aufgaben zum Thema Kalk.
Halogens
The compounds of halogens are - with the exception of astatine - widespread, can be encountered in nature and are versatile substances. This fact is taken up on this DVD in order to teach the students the chemistry of the halogens by illustrating their special qualities and explaining the correlation of their structure with their chemical properties. In the first part, an overview of the element group of halogens lays emphasis on the common as well as on the distinguishing characteristics of fluorine, chlorine, bromine and iodine. In a second part, the specific properties of bromine and iodine are presented. This topic is linked to the students‘ everyday experience on the one hand (bromine as a catalyst for reactolite sunglasses, iodine as an agent in medicine, etc.) on the one hand. As a rule, they are of a kind that can only be realized with difficulty, or high expenditure in the chemistry classroom. With the help of these experiments, students are introduced to the chemistry of the halogens in a way that enables them to draw conclusions on the basis of their observations.
Learn moreAcids and Bases
We can find acids and bases in every supermarket, some of them in our food, others in cleaning agents. In everyday products, acids and bases as well as acidic and alkaline reacting salts have extremely different functions. In food, acids are either present or added as flavouring agents such as citric acid, tartaric acid and acetic acid, as antioxidants such as ascorbic acid or generally as acidifiers, sequestrants (citric acid and tartaric acid) and preservatives (acetic acid).
Learn moreBasics of Chemistry II
When we take a closer look at substances, we discover that they consist of either one single element or of mixtures of several elements. Chemists therefore divide the world of substances into pure and mixed chemical substances. A pure substance is of homogeneous composition. Substance mixtures, however, consist of two or more pure substances. The many mixtures are subdivided not only into homogeneous and heterogeneous mixtures but depending on the respective aggregate states of their components, are classified into various groups of mixtures.
Learn moreBasics of Chemistry I
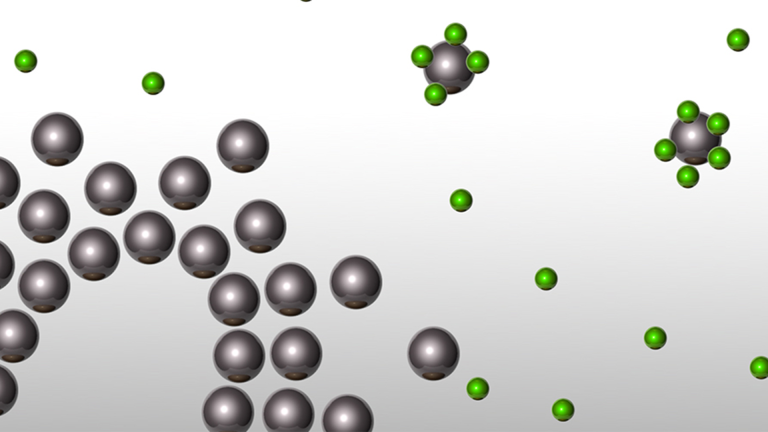
We are surrounded by objects and substances. We recognise objects that are to serve a specific purpose by their shapes. Similar objects may consist of different materials or substances. Substances, however, are independent of shapes and possess very specific properties. We are able to perceive many of these substances with our senses. For example, we can see, touch or smell them so as to be able to recognise them. Chemists are particularly interested in those substance characteristics that can be measured. On the basis of these measurable properties they can distinguish between substances, identify a specific substance or test it for special use. Models help us to understand phenomena. They depict only specific elements of our reality, thus presenting the world in a simplified way. The spherical particle model, for example, helps us to understand how a scent spreads all over the room or substances disperse in water.
Learn moreAluminium II
The metal aluminium is growing in importance because of its specific properties and manifold application possibilities. This DVD deals with the industrial production of aluminium as a raw material, its processing and the manufacturing of alloys for the finished product. Starting with the raw material aluminium oxide the functioning of an electrolytic cell is demonstrated and explained. Alumina, white and powdery, is melted with great expenditure of energy, and by means of electrolysis converted into aluminium with a degree of purity of 99.9%. As aluminium oxide would not melt before a temperature of over 2,000°C is reached, the mineral cryolite is used as a solvent. The various alloys change the properties of aluminium and are produced according to precise formulations. The alloy is cast into blocks and bars that serve as primary material for processing plants. The responsible handling of resources underscores the importance of recycling. Aluminium is resilient and versatile.
Learn more