55502616
Vergleich von ionischen und kovalenten Verbindungen
In 11 interaktiven Aufgaben wird Wissen zu ionischen und kovalenten Verbindungen vermittelt und anschließend abgefragt.
Das Medium bietet H5P-Aufgaben an, die ohne zusätzliche Software verwendbar sind.
Durch interaktive Aufgabentypen wird das audiovisuelle und interaktive Lernen einfach.
Lernen macht jetzt Spaß!
Included Tasks
- I Ionische und kovalente Verbindungen - Lückentext
- II Eine feste ionische Verbindung - Interaktive Aufgaben
- III Eine feste molekulare kovalente Verbindung - Interaktive Aufgaben
- IV Eigenschaften - Gitterstruktur - Interaktive Aufgaben
- V Die Eigenschaften von ionischen Verbindungen - Interaktive Aufgaben
- VI Die Eigenschaften von kovalenten Verbindungen - Interaktive Aufgaben
- VII Eine Verbindung-Quiz - Interaktive Aufgaben
- VIII Kovalent oder ionisch? - Interaktive Aufgabe
- IX Löslichkeit in organischen Lösungsmitteln - Interaktive Aufgaben
- X Warum leiten feste ionische Verbindungen keinen Strom - Interaktive Aufgaben
- XI Kovalente Moleküle - Interaktive Aufgaben
Curriculum-centred and oriented towards educational standards
Matching
Halogens
The compounds of halogens are - with the exception of astatine - widespread, can be encountered in nature and are versatile substances. This fact is taken up on this DVD in order to teach the students the chemistry of the halogens by illustrating their special qualities and explaining the correlation of their structure with their chemical properties. In the first part, an overview of the element group of halogens lays emphasis on the common as well as on the distinguishing characteristics of fluorine, chlorine, bromine and iodine. In a second part, the specific properties of fluorine and chlorine are presented. This topic is linked to the students‘ everyday experience (fluorine as a protection against caries, chlorine as a disinfectant, etc.) on the one hand. On the other hand, the DVD presents carefully selected experiments. As a rule, they are of a kind that can only be realized with difficulty, or high expenditure in the chemistry classroom. With the help of these experiments, students are introduced to the chemistry of the halogens in a way that enables them to draw conclusions on the basis of their observations.
Noble Gases
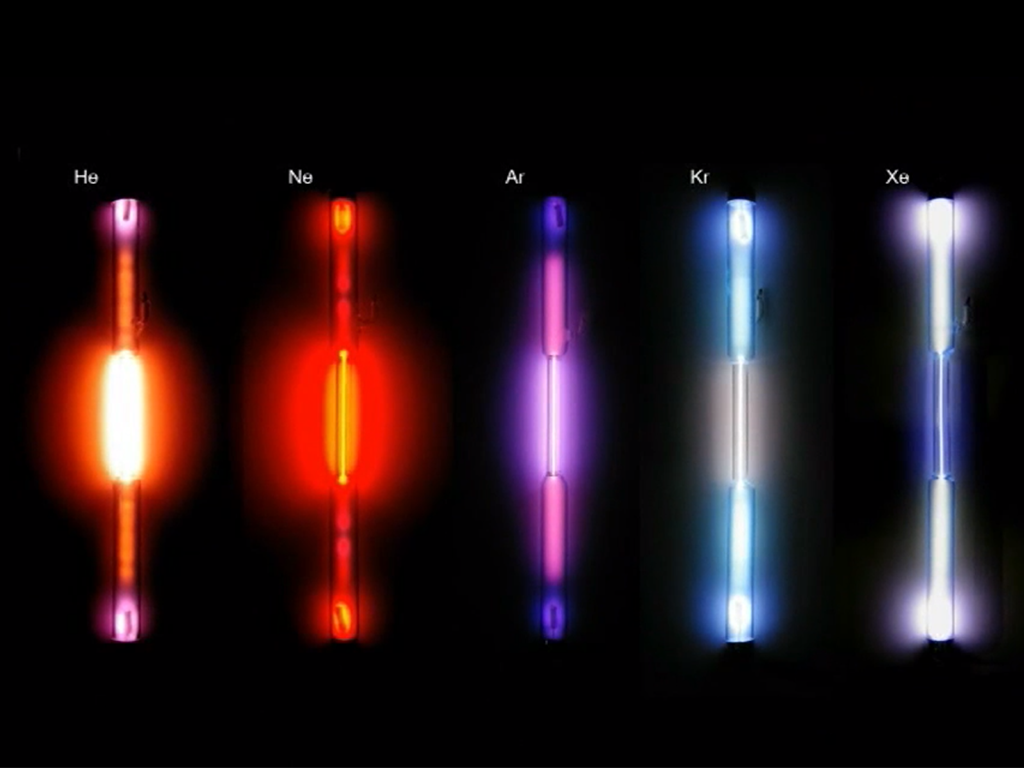
Xenon, Helium, Neon, Argon, Krypton and the radioactive Radon belong to the noble gases. These form the family of noble gases as the elements of the eighth group of the periodic table. All of them are colourless and odourless, non-inflammable and non-toxic. Their most striking chemical property is their inertness. This can be explained by their electron arrangement, termed noble gas configuration and represents a particularly stable and therefore low-energy state. The noble gases are to be found in scant amounts in our air from which they are also distilled. Helium is mainly extracted from natural gas. In everyday life, we encounter noble gases for example as shielding, filling or buoyant gases and in fluorescent tubes. The shell model describes the structure of the atoms. It is based on the distribution of electrons in restricted areas at a fixed distance around the core of the atom.
Halogens
The compounds of halogens are - with the exception of astatine - widespread, can be encountered in nature and are versatile substances. This fact is taken up on this DVD in order to teach the students the chemistry of the halogens by illustrating their special qualities and explaining the correlation of their structure with their chemical properties. In the first part, an overview of the element group of halogens lays emphasis on the common as well as on the distinguishing characteristics of fluorine, chlorine, bromine and iodine. In a second part, the specific properties of bromine and iodine are presented. This topic is linked to the students‘ everyday experience on the one hand (bromine as a catalyst for reactolite sunglasses, iodine as an agent in medicine, etc.) on the one hand. As a rule, they are of a kind that can only be realized with difficulty, or high expenditure in the chemistry classroom. With the help of these experiments, students are introduced to the chemistry of the halogens in a way that enables them to draw conclusions on the basis of their observations.